When to Refer for a Lymphedema Pump: A Clinical Guide | 2026
The clinical question of when to escalate from compression garments to a pneumatic compression device (PCD) has gotten more interesting in 2026. The therapy itself hasn't changed — segmental and non-segmental pumps still do exactly what they've always done, and the patient populations who benefit are well-described in the literature. What's changed is the regulatory and documentation environment: nationwide prior authorization for segmental devices took effect April 13, 2026, and CMS's 2024 Fee-for-Service Supplemental Improper Payment Data placed the PCD improper payment rate at 61.5% — driven primarily by insufficient documentation. Together, those two facts mean that the difference between a referral that gets approved and one that gets denied is mostly upstream of the supplier, in the prescriber's note.
This guide is for the clinicians who make the referral decision: primary care, oncology, vascular and lymphatic specialists, NPs and PAs in compression-heavy practices, and the certified lymphedema therapists who often surface the question first. It walks through the clinical profile that warrants a PCD evaluation, the four-week conservative therapy gateway every Medicare claim depends on, how to choose between E0650, E0651, and E0652 codes, what the 2026 prior authorization process actually looks like, and the documentation patterns that hold up in audit.
Pumps are a genuinely valuable tool for the right patient. They're also, statistically, the lymphedema item Medicare denies most often. Both can be true, and getting the referral right is what reconciles them.
PCDs Are Not LTA Items: The First Distinction That Matters
Pneumatic compression devices are covered under the traditional DME benefit governed by National Coverage Determination 280.6 and Local Coverage Determination L33829 — not under the Lymphedema Treatment Act benefit category established in 2024. This matters operationally because:
PCDs require a four-week trial of conservative therapy before coverage; LTA garments do not.
PCDs E0651 and E0652 require prior authorization as of April 13, 2026 nationwide; LTA garments do not.
PCD coverage hinges on clinical findings of severity beyond the diagnosis itself; LTA garment coverage hinges primarily on the diagnosis.
PCDs and LTA garments are billed under different code sets (E0650/E0651/E0652 vs. the Axxxx LTA codes) and audited under different rules.
Practically: the LTA changes don't affect PCD prescribing. PCD policy operates on its own track, and it's stricter. A patient who qualifies for compression garments under the LTA does not automatically qualify for a PCD — the threshold is meaningfully higher.
For broader background on lymphedema management at MCB DME, see mcbdme.com/lymphedema.
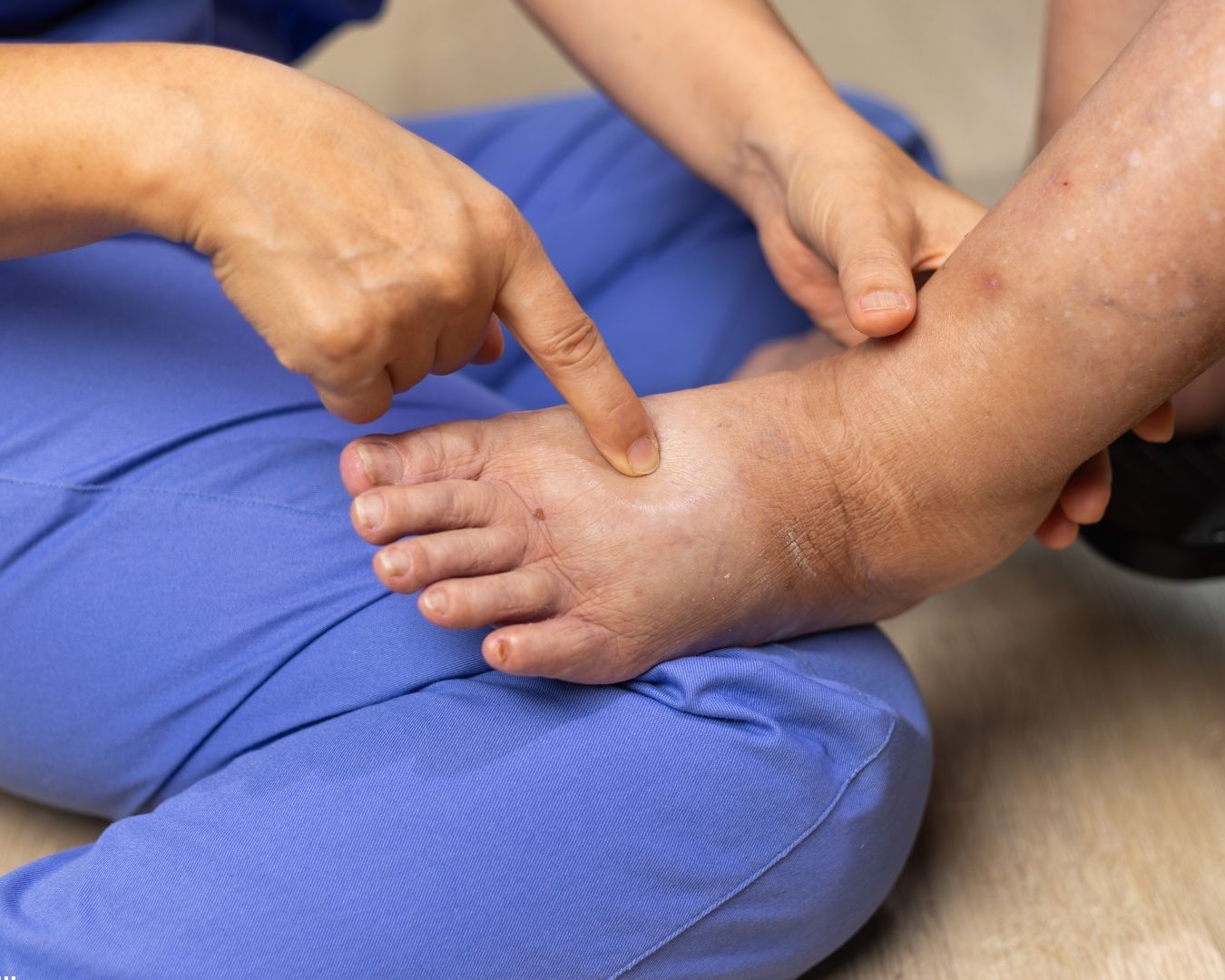
The Clinical Profile That Warrants a PCD Referral
A pneumatic compression device is appropriate when conservative therapy alone isn't adequately controlling the patient's lymphedema or, in select cases, chronic venous insufficiency with venous stasis ulcers. The clinical picture that most reliably supports a PCD referral includes one or more of these features:
Persistent, chronic, severe lymphedema that has not responded to consistent compression and decongestive therapy.
Documented failure of a four-week trial of compression bandaging or graduated compression garments combined with exercise and limb elevation.
Clinical findings of severity in the affected limb or region — marked hyperkeratosis with hyperplasia and hyperpigmentation, lymphorrhea (skin weeping), papillomatosis, recurrent cellulitis, or functional impairment.
Lymphedema extending beyond the limb onto the chest, trunk, or abdomen, where standard compression garments alone cannot deliver adequate therapy.
Patient circumstances that compromise garment compliance — but where pump therapy is realistic and supported.
Chronic venous insufficiency with venous stasis ulcers that have failed a six-month trial of conservative therapy including compression and appropriate wound care.
Patients who are not appropriate PCD candidates include those with edema unrelated to lymphedema or CVI (e.g., heart failure–related edema, lipedema without lymphedema component, idiopathic edema), patients showing meaningful improvement with conservative therapy, and patients with active untreated DVT, severe peripheral arterial disease, or other contraindications to external compression.
It's worth saying directly: lipedema without an established lymphedema component does not meet Medicare's PCD criteria, even though the symptom presentation can feel similar. If both conditions coexist (lipo-lymphedema), the lymphedema component must be diagnosed and documented as such for coverage to apply.
The Four-Week Conservative Therapy Gateway
This is the documentation point that fails most PCD claims. The four-week trial is not optional, and "the patient has tried compression" doesn't satisfy the requirement. Per the LCD, the four-week trial must include all of the following, documented in the medical record:
Regular and compliant use of an appropriate compression bandage system or graduated compression garment. Generic "wears stockings" language doesn't qualify — the record should specify the garment type, fit verification, and adherence.
Exercise appropriate to the patient's limb and condition.
Elevation of the affected limb.
Treating practitioner determination that there has been no significant improvement, or that significant symptoms remain after the trial.
The record should also include objective measurements — limb circumference at standard reference points, volume measurements where available, photographic documentation of skin changes — both at the start of the conservative trial and at its conclusion. "No improvement" without measurements is the single most common reason the four-week trial documentation gets rejected on audit.
Where the conservative therapy was directed by a certified lymphedema therapist (which is common and clinically appropriate), the CLT's evaluation and progress notes are valuable supporting evidence. But the diagnosis and the determination that conservative therapy failed must be documented in the prescribing clinician's own note — MD, DO, NP, PA, or CNS. CLT documentation alone, even when countersigned, does not meet Medicare's prescriber-level documentation requirement. (DPM/podiatry is specifically excluded from PCD prescribing under the LCD, regardless of state scope.)

Choosing the Right PCD Code: E0650 vs E0651 vs E0652
The HCPCS distinctions matter clinically and financially. Each code corresponds to a specific device class, and Medicare's coverage criteria differ:
E0650 — Non-segmental pneumatic compressor (single chamber)
A basic pump that inflates a single-chamber appliance uniformly. Appropriate for patients with mild to moderate extremity lymphedema who haven't responded to conservative therapy. Lowest cost, simplest device, and the entry point for most PCD prescriptions.
E0651 — Segmental pneumatic compressor without calibrated gradient pressure
A more sophisticated pump with multiple chambers that inflate sequentially to mimic the milking action of manual lymphatic drainage. Most prescriptions for limb lymphedema land here. Requires prior authorization as of April 13, 2026.
E0652 — Segmental pneumatic compressor with calibrated gradient pressure
The most advanced — and most restrictively covered — class. Per Medicare policy, E0652 is covered only when:
The patient has lymphedema extending onto the chest, trunk, or abdomen beyond the limits of a standard compression sleeve, and
The patient has had a four-week trial of an E0650 or E0651 device with documented failure to improve, with pre- and post-measurements.
Coverage of E0652 for lymphedema of the extremities alone is excluded, even when other criteria are met — that's a specific carve-out in NCD 280.6 and L33829. Trying to bypass this with "unique characteristics that prevent satisfactory treatment with E0651" requires highly specific clinical justification, and Noridian explicitly notes that listing only "unique characteristics" without supporting detail will result in denial. E0652 requires prior authorization as of April 13, 2026.
The major manufacturers in MCB DME's referral mix span all three classes:
BioCompression Systems — long-established, broad device line spanning E0650 through E0652.
Tactile Medical — Flexitouch Plus (E0652) and Nimbl (E0651) — strong evidence base in head-and-neck and trunk lymphedema for the Flexitouch.
Lympha Press — full PCD line with strong patient education infrastructure.
Koya Medical — Dayspring, a wearable, mobile E0651-class device.
Selection between manufacturers within a code class is typically a function of patient anatomy, lifestyle, mobility, prior experience, and clinical fit. MCB DME coordinates trial fittings and manufacturer support directly so the prescriber doesn't have to.
For broader compression therapy resources, see mcbdme.com/compression.
The 2026 Prior Authorization Reality
CMS's Master List for Prior Authorization added E0651 and E0652 to the nationwide PA list effective April 13, 2026. That means before a segmental PCD is delivered to a Medicare beneficiary, the supplier must submit a PA request with full documentation, and Medicare must affirmatively approve it.
The good news: real-world experience with PCD PA has been notably less painful than other PA programs. Standard decisions typically arrive within 10 business days, often faster. Expedited decisions (when delay would jeopardize the patient) can arrive within 2 business days. The supplier handles the PA submission; the prescriber's role is providing the documentation that supports the request.
A complete PA submission includes:
Standard Written Order (SWO) signed by the prescribing clinician, specifying the HCPCS code, quantity, and start date.
Treating practitioner's medical records documenting the lymphedema diagnosis with ICD-10 code, severity findings, the four-week conservative trial details, and pre/post measurements.
Face-to-face encounter with the prescribing clinician within six months of the order date, with the encounter note documenting the clinical findings supporting PCD necessity.
For E0652: documentation of the trunk/chest/abdomen extension or documented failure of an E0651 device with pre and post measurements.
For replacement or upgrades: clinical response to prior PCD treatment, including measurements.
If a PA request is denied, the supplier can resubmit with additional supporting documentation as many times as needed. Most denials are correctable; few are categorical.
Documentation Patterns That Survive Audit
CMS's 61.5% improper payment rate for PCDs in 2024 was driven 62.9% by insufficient documentation and 37.1% by failure to meet medical necessity. Both are upstream of the device — they live in the prescriber's note. Patterns that consistently hold up in audit:
Specific measurements at specific time points. "Limb circumference 41 cm at the mid-bicep at initial evaluation, 39 cm after six weeks of compression therapy, no further reduction over four additional weeks" is dramatically more defensible than "minimal improvement with compression."
Named clinical findings. "Stage II lymphedema with marked hyperkeratosis and recurrent cellulitis (two episodes in past 12 months)" survives audit; "chronic swelling" doesn't.
Conservative therapy specifics. Documented compression garment type and fitting, documented exercise prescription, documented elevation regimen, with adherence and outcome notes — not "patient instructed in compression."
Functional impairment language where applicable. "Limb heaviness limiting fine motor tasks" or "weight of edematous arm causing shoulder pain restricting overhead motion" connects the swelling to a functional consequence, which strengthens medical necessity.
Coordination notes with the lymphedema therapist. A short reference to the CLT's findings — "CLT evaluation by [therapist] on [date] documented [findings] and recommended PCD evaluation given [reason]" — uses the CLT's clinical work without making it the primary diagnostic note.
Clinical response after PCD initiation. For continued coverage and audit defense, document the patient's response — measurements, symptoms, function — within a few weeks of starting therapy.
The single highest-leverage habit is taking measurements at consistent reference points at consistent intervals. It transforms the chart from "this seems bad" to "here is the trajectory."
Common Mistakes That Drive PCD Denials
From the operational pattern of denied claims and Medicare's own published improper payment analysis:
No documented four-week trial. Either the trial wasn't done, or it was done but not adequately documented in the prescriber's own note.
E0652 prescribed for limb lymphedema without trunk involvement. This is a categorical exclusion under L33829, regardless of how severe the limb is.
Diagnosis of "lymphedema" without supporting clinical findings. Coverage requires not just the diagnosis but the documented severity.
Conservative therapy successful but PCD prescribed anyway. If the patient improved with conservative therapy, the PCD is not medically necessary by definition.
Edema from causes other than lymphedema or CVI with ulcers. Cardiac edema, lipedema without lymphedema, idiopathic edema — none qualify.
Missing face-to-face encounter within six months of the order date.
Podiatrist-issued PCD orders. DPMs are not permitted to order PCDs under L33829, regardless of state scope of practice.
How MCB DME Supports the PCD Referral
MCB DME is a Medicare-enrolled DMEPOS supplier serving northern New Jersey and the broader region. For PCD referrals specifically, this means:
End-to-end PA management. Once a referral arrives, MCB DME assembles the prior authorization submission, verifies documentation completeness with the referring practice before submitting, and tracks the determination through to delivery.
Manufacturer coordination. Direct relationships with BioCompression, Tactile Medical, Lympha Press, and Koya Medical for device selection, fitting, and patient training. We help match the patient profile to the device, not the other way around.
BioCompression travel letter for patients flying with PCD components — available to any MCB DME PCD patient via mcbdme.com/contactus.
Documentation feedback. Where a referral arrives with documentation gaps that would generate a PA denial, our intake team flags them back to the referring practice before the submission goes out.
Continued patient support. Device training, supply replacement, garment troubleshooting, and clinical response documentation support after delivery.
Prescription forms and provider resources at mcbdme.com/providers.
Frequently Asked Questions
Does the four-week conservative trial have to be exactly four weeks?
The LCD specifies a minimum of four weeks. Longer is fine — and for patients in active lymphedema therapy, the conservative trial often runs six weeks or more by the time a PCD is being considered. The minimum threshold is what's audit-relevant.
Can the patient's CLT direct the conservative therapy trial?
Yes, and that's clinically appropriate. The CLT-directed trial is excellent supporting evidence. The treating practitioner must still document, in their own note, the diagnosis, the trial elements, and the determination that conservative therapy failed.
What if the patient already has a compression garment under the LTA — does that count as conservative therapy?
Yes. A documented period of consistent garment use at appropriate compression class, with adherence verified and outcomes measured, satisfies the compression component of the four-week trial. This is one place where the LTA and PCD policies usefully connect.
Are PCDs covered by Medicare Advantage plans?
MA plans must cover everything Original Medicare covers, including PCDs that meet NCD 280.6 criteria. In practice, expect plan-specific networks, additional PA requirements, and submission portals. MCB DME verifies MA plan requirements at intake.
Is prior authorization required for E0650?
Currently, prior authorization at the national level applies to E0651 and E0652 (effective April 13, 2026). E0650 is not on the national PA list at this time, though individual MACs may apply additional review. Documentation requirements remain identical regardless of PA status.
Can a patient have a PCD and LTA-covered compression garments simultaneously?
Yes — they're complementary, billed under different benefit categories, and clinically often used together. The PCD provides intermittent active compression; the garment provides continuous gradient compression between pump sessions and overnight.
What about the 24-hour wearable Koya Dayspring — same coverage rules?
Yes. The Dayspring is classified as an E0651 segmental pneumatic compression device for Medicare purposes and falls under the same NCD 280.6 coverage criteria, four-week conservative trial requirement, and prior authorization framework.
How long does the PA decision take?
Standard decisions typically arrive within 10 business days, often 5–7. Expedited decisions (when delay would jeopardize the patient) can arrive within 2 business days. Most denials are correctable on resubmission with additional documentation.
Key Takeaways
PCDs sit under a separate Medicare benefit category from LTA garments, with a four-week conservative therapy gateway, severity findings requirements, and (as of April 13, 2026) nationwide prior authorization for E0651 and E0652.
The clinical decision to refer for a PCD hinges on documented failure of conservative therapy plus clinical findings of severity — not on the lymphedema diagnosis alone.
HCPCS code selection matters: E0650 for basic non-segmental, E0651 for most limb lymphedema, E0652 reserved for lymphedema extending onto chest/trunk/abdomen or after documented E0651 failure.
Insufficient documentation drives most PCD denials — specific measurements at specific time points, named clinical findings, and concrete conservative therapy details survive audit.
The diagnosis and the determination that conservative therapy failed must live in the prescribing clinician's own note, with MD/DO/NP/PA/CNS authorship — CLT documentation supports but does not substitute.
A knowledgeable DMEPOS supplier handles the PA submission, manufacturer coordination, and post-delivery support, leaving the prescriber to focus on the clinical decision and documentation.
Next Steps
If you have a patient on conservative therapy whose lymphedema isn't adequately controlled — or who has clinical findings of severity that conservative therapy alone won't address — three concrete actions move the referral forward cleanly:
Document the four-week conservative trial in your own progress note, with specific compression therapy details, exercise and elevation prescriptions, measurements at start and conclusion, and your clinical determination of inadequate response.
Identify which HCPCS code class fits the patient — E0650 for basic, E0651 for segmental (most lymphedema), E0652 only for trunk/chest/abdomen extension or post-E0651 failure.
Refer to MCB DME with the prescription and supporting documentation. We handle prior authorization, manufacturer selection, fitting, training, and ongoing support.
For prescription forms, referral resources, and direct contact with the MCB DME team, visit mcbdme.com/providers. For broader lymphedema management resources or to reach our intake and PA team directly, see mcbdme.com/contactus.
Related Resources:
MCB DME is a durable medical equipment provider based in Hawthorne, New Jersey, specializing in lymphedema management, compression therapy, diabetic footwear, arterial pumps, and bracing and prosthetics. MCB DME is a Medicare-enrolled DMEPOS supplier and bills Medicare, Medicaid, and most commercial insurance plans directly for both LTA-covered compression items and traditional DME pumps.
Disclaimer: This article is for educational purposes only and does not constitute legal, billing, or compliance advice. Coverage criteria, prior authorization requirements, documentation standards, and HCPCS coding are subject to change; always verify current CMS, DME MAC, and payer policies for guidance specific to your practice. MCB DME does not guarantee insurance approval for specific devices or patients.
